Lora Uses, Dosage, Side Effects and more
Lora is a short-acting tranquilizer of the benzodiazepine group. It is an agonist at benzodiazepine receptors in the CNS. It exerts a depressant action on the CNS, which may be mediated by potentiating the inhibitory actions of GABA (gamma amino butyric acid) in the CNS. This will result in diminution of the ascending activating systems, particularly the serotonergic and noradrenergic pathway from brain stem or the mid-brain to the cerebral cortex.
The effect of lorazepam in GABA-A receptors produces an increase in the frequency of opening of the chloride ion channel. However, for its effect to generate, the neurotransmitter is required. The anticonvulsant properties of lorazepam are thought to be related to the binding to voltage-dependent sodium channels in which the sustained repetitive firing gets limited by the slow recovery of sodium channels due to the benzodiazepine effect.
The effect of lorazepam seems to be very compartmental which was observed with a different generation of sleepiness and a dizziness effect.
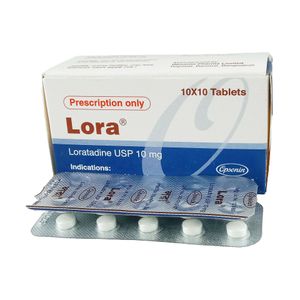
| Attribute | Details |
|---|---|
| Trade Name | Lora |
| Availability | Prescription only |
| Generic | Lorazepam |
| Lorazepam Other Names | Loracepam, Lorazepam, o-Chlorooxazepam, o-Chloroxazepam |
| Related Drugs | sertraline, trazodone, escitalopram, fluoxetine, alprazolam, duloxetine, hydroxyzine, Lexapro, amitriptyline, ondansetron |
| Weight | 10mg, 5mg/5ml, 2mg, |
| Type | Tablet, Injection, Oral Suspension |
| Formula | C15H10Cl2N2O2 |
| Weight | Average: 321.158 Monoisotopic: 320.011932988 |
| Protein binding | Reports indicate that 85% of lorazepam administered dose is protein bound. |
| Groups | Approved |
| Therapeutic Class | Benzodiazepine hypnotics, Benzodiazepine sedatives |
| Manufacturer | Abs Remedies, Yaher Pharma, Orchid Pharma, Opsonin Pharma Ltd |
| Available Country | India, Bangladesh, |
| Last Updated: | January 7, 2025 at 1:49 am |
Uses
Lora is used for the treatment of anxiety states; including anxiety associated with phobic & obsessional states, psychosomatic, organic or psychotic illness, insomnia associated with anxiety, nervousness, restlessness, nausea and vomiting related to chemotherapy and anticonvulsants, and as a premedicant before dental or general surgery or prior to investigative procedures where there may be discomfort. Lora is not recommended in anxiety states in children but may be used as premedicant before surgery at a dose of 0.05 mg/Kg in children aged 5 to 13 years. The dosage of Lora tablet should be increased gradually when needed to avoid adverse effects. When higher dosage is used, the evening dose should be increased before the daytime.
Lora is also used to associated treatment for these conditions: Agitation, Alcohol Withdrawal Delirium, Alcohol Withdrawal Syndrome, Anxiety, Anxiety Disorders, Catatonia, Chemotherapy-Induced Nausea and Vomiting (CINV), Delirium, Insomnia, Muscle Spasms, Panic Disorder, Refractory seizure disorders, Preanesthetic medication therapy
How Lora works
Lora allosterically binds on the benzodiazepine receptors in the post-synaptic GABA-A ligand-gated chloride channel in different sites of the central nervous system (CNS). This binding will result in an increase on the GABA inhibitory effects which is translated as an increase in the flow of chloride ions into the cell causing hyperpolarization and stabilization of the cellular plasma membrane.
According to the binding site of lorazepam, we can observe different activities as the binding in the amygdala is known to help mainly in anxiety disorders while the binding in the cerebral cortex helps in seizure disorders.
Dosage
Oral-
Frequency of administration and duration of therapy should be individualized according to patient's response. The usual range is 2 to 6 mg/day given in divided doses, the largest dose being taken before bedtime, but the largest dosage may vary from 1 to 10 mg/day. For anxiety, most patients require an initial dose of 2 to 3 mg/day given b.i.d. or t.i.d. For elderly or debilitated patients, an initial dosage of 1 to 2 mg/day in divided doses is recommended, to be adjusted as needed and tolerated. For insomnia 1 to 2 mg before bedtime and as premedicant 1 to 2 mg the night before surgery and 1 to 2 hour before surgery.
Injection-
Acute anxiety:
- Adult:IV/IM: 25-30 mcg/kg repeated 6 hrly if necessary. Give IV inj at a rate of not more than 2 mg/min into a large vein.
- Child:<12 yrNot recommended.
- Elderly:Half the usual adult dose or less.
Premedication in surgery:
- Adult:50 mcg/kg given pre-op over 30-45 min (IV) or 60-90 min (IM).
- Child:<12 yrNot recommended.
- Elderly:Half the usual adult dose or less.
Status epilepticus:
- Adult:4 mg as a single dose, into a large vein at a rate not more than 2 mg/min. May be repeated once after 10-15 min if seizures recur.
- Child:2 mg as single dose.
- Elderly:Half the usual adult dose or less.
Injection: Dilute with an equal volume of compatible diluent (e.g. sterile water for inj, NaCl 0.9% inj, dextrose 5% inj).
Oral concentrate solution: Dose should be added to 30 mL or more of diluent (e.g. water, juice, carbonated or soda-like beverage) or to semi-solid food (e.g. applesauce, pudding).
How Long Does It Take to Work?
How Long Does It Take to Work? see here Lora
Side Effects
Adverse reactions are usually observed at the beginning of therapy and generally disappeared on continued medication or upon decreasing the dose. The most frequent adverse reactions of Lora are sedation followed by dizziness, weakness and unsteadiness. Less frequent adverse reactions are disorientation, depression, nausea, change in appetite, headache, sleep disturbance, agitation, dermatological symptoms, eye-function disturbance, together with various gastrointestinal symptoms and autonomic manifestations.
Toxicity
The LD50 observed by oral administration in a mouse is of 1850 mg/kg. When an overdose administration is registered, signs of CNS and respiratory depression are rapidly observed. An overdose stage can result in profound sedation, deep respiratory depression, coma, and death. When overdose is observed, it is recommended to administer emergency symptomatic medical support with attention to produce an increase in lorazepam elimination.
There is no evidence of carcinogenicity nor mutagenicity. At doses higher than 40 mg/kg there is evidence of fetal resorption and increase in fetal loss.
Precaution
In patients with depression accompanying anxiety, a possibility for suicide should be borne in mind. For elderly or debilitated patients, the initial daily dosage should not exceed 2 mg in order to avoid over sedation. The usual precautions for treating patients with impaired renal or hepatic function and with acute and chronic pulmonary insufficiency should be observed. Use of Lora for prolonged period and gastric patients requires caution and there should be frequent monitoring for symptoms of upper gastrointestinal disease. Machineries should be avoided during taking Lora.
Interaction
Lora may potentiate the central depressant effect when combined with other drugs with central depressant action. Such drugs include alcohol, general anesthetics, tricyclic antidepressants and monoamine oxidase inhibitors.
Food Interaction
- Avoid alcohol.
- Limit caffeine intake.
- Take with food.
[Moderate] GENERALLY AVOID: Alcohol may potentiate some of the pharmacologic effects of CNS-active agents.
Use in combination may result in additive central nervous system depression and
MANAGEMENT: Patients receiving CNS-active agents should be warned of this interaction and advised to avoid or limit consumption of alcohol.
Ambulatory patients should be counseled to avoid hazardous activities requiring complete mental alertness and motor coordination until they know how these agents affect them, and to notify their physician if they experience excessive or prolonged CNS effects that interfere with their normal activities.
Drug Interaction
Major: acetaminophen / hydrocodoneModerate: aripiprazole, diphenhydramine, duloxetine, escitalopram, pregabalin, quetiapine, sertraline, cetirizineMinor: metoprololUnknown: amphetamine / dextroamphetamine, aspirin, omega-3 polyunsaturated fatty acids, polyethylene glycol 3350, esomeprazole, levothyroxine, acetaminophen, cyanocobalamin, ascorbic acid, cholecalciferol
Disease Interaction
Major: acute alcohol intoxication, closed-angle glaucoma, drug dependence, renal/liver disease, respiratory depression, seizures, prolonged hypotension, prematurityModerate: depression, obesity, paradoxical reactions, upper GI disease
Volume of Distribution
The reported volume of distribution of lorazepam is 1.3 L/kg. It is important to mention that due to the lipophilicity of lorazepam, it does not redistribute as fast in the brain.
Elimination Route
Readily absorbed with an absolute bioavailability of 90% when given orally. When intramuscularly administered a dose of 4 mg, lorazepam is completely and rapidly absorbed and achieves a maximal serum concentration of 48 ng/ml in 15-30 minutes. When administered orally, the time to attained maximum concentration is observed to be of 2 hours.
Half Life
When administered parentally, the registered half-life of lorazepam is of 14 hours.
Clearance
In vivo studies with lorazepam have shown a clearance rate of 5.8 ml.min/kg.
Elimination Route
When a single 2 mg oral dose is given to healthy subjects, 88% of the administered dose is recovered in urine and 7% was recovered in feces. From the excreted dose in urine, the major form is the glucuronide version that represents 74% while only 0.3% of the dose is recovered as unchanged lorazepam.
Pregnancy & Breastfeeding use
Safety of Lora in pregnancy has not been established. Because the use of this drug is rarely a matter of urgency, the use of Lora during this period should almost always be avoided.
It is not known whether oral Lora is excreted in human milk like other benzodiazepine tranquilizers. As a general rule, nursing should not be undertaken while a patient is on a drug since many drugs are excreted in human milk.
Contraindication
Lora should not be given to patients with a previous sensitivity to benzodiazepines or with acute narrow-angle glaucoma and myasthenia gravis.
Special Warning
Debilitated patient: Half the usual adult dose or less.
Renal Impairment: May require reduced doses.
Mild to Moderate Hepatic Impairment: May require reduced doses.
Severe Hepatic Impairment: Contraindicated.
Acute Overdose
Overdosage of Lora is usually manifested by varying degrees of central nervous system depression ranging from drowsiness to coma. In mild cases, symptoms include drowsiness, mental confusion and lethargy. In more serious cases and especially when other drugs or alcohol were ingested, symptoms may include ataxia, hypotonia, hypotension, hypnotic states (stages I to III), coma.
Storage Condition
Tablet: Store between 20-25°C.
Injection or oral concentrate solution: Store between 2-8°C. Do not freeze. Protect from light.
Innovators Monograph
FAQ
What is Lora used for?
It is used to treat anxiety disorders, trouble sleeping, severe agitation, active seizures including status epilepticus, alcohol withdrawal, and chemotherapy-induced nausea and vomiting. It can be taken to help you relax before an operation or other medical or dental treatment.
How safe is Lora?
Lora is a safe and effective medication when used as directed.
How does Lora work?
Lora works by increasing the levels of a calming chemical, gamma-amino-butyric-acid (GABA), in your brain.
What are the common side effects of Lora?
Common side effects of Lora are include:
- drowsiness
- dizziness
- tiredness
- weakness
- dry mouth
- diarrhea
- nausea
- changes in appetite
- restlessness or excitement
- constipation
- difficulty urinating
- frequent urination
- blurred vision
- changes in sex drive or ability
Is Lora safe during pregnancy?
Lora should be avoided during pregnancy.
Is Lora safe during breastfeeding?
Evidence from nursing mothers indicates that Lora does not cause any adverse effects in breastfed infants with usual maternal dosages.
Can I drink alcohol with Lora?
Alcohol can increase the nervous system side effects of Lora such as dizziness, drowsiness, and difficulty concentrating.
When should be taken of Lora?
Lora may be taken with or without food. Take with food if you experience an upset stomach. Lora may be taken every day at regular times or on an as needed.
How often can I take Lora?
Adults, one capsule once a day in the morning. Dose is based on the total daily dose of Lora tablets, which you take three times a day in equally divided doses.
How long does Lora take to work?
Lora tablets and liquid start to work in around 20 to 30 minutes. The full sedating effect lasts for around 6 to 8 hours.
How long does Lora stay in my system?
Lora may be detectable in urine samples for up to six days, and some of the metabolites may be detectable for even longer (nine days). If regularly abused, urinalysis may reveal the presence of Lora after a week or longer.
How long can I stay on Lora?
It's not recommended to use Lora for longer than 4 weeks. The most common side effect is feeling sleepy (drowsy) during the daytime.
Who should not take Lora?
If you have or have ever had seizures; or lung, heart, or liver disease you should not take Lora. Tell your doctor if you are pregnant, plan to become pregnant, or are breastfeeding.
What happen If I missed Lora?
If you miss a dose of Lora, take the missed dose as soon as you remember it. If it is almost time for your next dose, skip the missed dose and continue with your dosing schedule. Do not double up your dose of Lora to make up for the missed dose.
What happens if I overdose of Lora?
Overdose symptoms may include extreme drowsiness, confusion, slurred speech, feeling restless, muscle weakness, loss of balance or coordination, feeling light-headed, slow heartbeats, weak or shallow breathing, or coma. If you think you or someone else may have overdosed on: Lora (Ativan), call your doctor or the Poison Control center.
Can Lora affects my heart ?
With an Lora addiction comes a variety of health issues, such as heart problems. Lora affects the heart by slowing your heart rate and lowering your blood pressure.
Can Lora affect my kidneys?
Lora can also damage liver and kidney function.
Can Lora affect my liver?
Clinically apparent liver injury from Lora is extremely rare.
Does Lora cause memory loss?
Lora slows down the functions of the brain and body.
Can I cut Lora in half?
Swallow the tablet whole. Do not crush, break, or chew it.



